Grünenthal has published its Responsibility Report 2025/26, showcasing what the company delivered in 2025 across environmental, social and governance topics.
Grünenthal is a global leader in pain management.
Grünenthal is a global leader in pain management and related diseases. As a science-based, privately-owned pharmaceutical company, we have a long track record of bringing innovative treatments and state-of-the-art technologies to patients worldwide. Our purpose is to change lives for the better, and innovation is our passion. We are focusing all our activities and efforts on working towards our vision of a world free of pain.

Grünenthal at a glance – experience our virtual company showcase
Read about our key objectives and activities, as well as our recent business development highlights and financial performance. Get insights into our leadership team, strategy and financials, R&D & Pipeline, Product portfolio, Reliable Supply, Environmental & Societal Governance, and our Company culture and purpose.
Grünenthal Stories

Fascination for research and innovation
Grünenthal pursues its vision of a World Free of Pain through extensive research and development. Experts from a variety of disciplines constantly search for new substances that could become active ingredients of innovative pain therapies – driven by the needs of millions of people who suffer from chronic pain around the world.
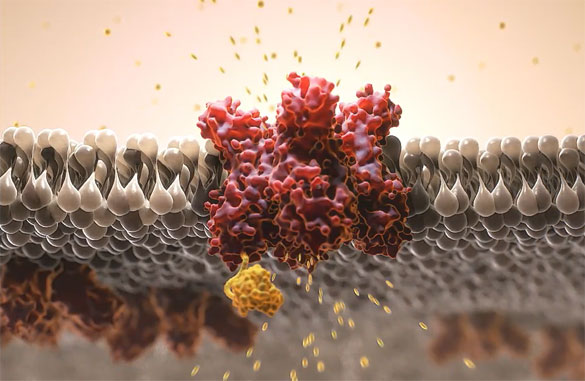
First-In-Human Trial with NaV 1.8 inhibitor

Grünenthal Report 2025/26
Grünenthal is driven by a clear and ambitious vision: a World Free of Pain. In 2025, we made significant progress in advancing innovative medicines for patients living with pain worldwide.
The Grünenthal Report is a way we share updates on our collective projects, priorities, and progress. It provides an overview of our continued efforts to move closer to our vision.

Responsibility Report 2025/26

Careers
Working at Grünenthal means experiencing the impact you can have on the results we achieve and the lives of the patients we serve.
Join forces.
Make an impact.
Innovate for a world free of pain.
Visit our careers website


















