

Where we come from
Although firmly rooted in Germany, today we are very much a growing global business. As an integrated research and development company, we have a strong track record of bringing effective pain treatments and state-of-the-art technologies to patients. Our high level of investment in R&D is testament to our unwavering commitment to innovation.


1947 Penicillin

1957 Thalidomide

1968 International expansion

1977 Tramal®

2001 Transtec®

2003 Zaldiar®

2007 Versatis®

2010 Palexia® retard

2013 Andrómaco
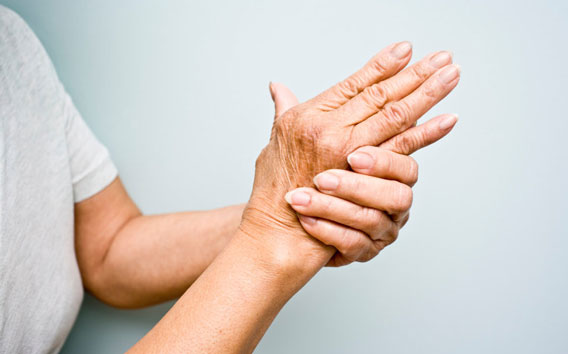
2014 Arcoxia®

2017 Zomig®

2018 Nexium® and Vimovo®

2018 Qutenza®
2020
2021
Grünenthal celebrates its 75th anniversary. It is also the 60th year since the market withdrawal of Thalidomide.
With the European rights (except Spain and the U.K.) for the lipid-lowering agent Crestor®, as well as the Swiss company Mestex AG, with its innovative investigational medicine RTX (resiniferatoxin) for the intra-articular treatment of pain associated with osteoarthritis of the knee, we complete two further significant acquisitions.

2022 Nebido®

2023 Grünenthal Meds

2024 Acquisition of Valinor Pharma

2025

2026
M-N/A-HQ-04-25-0010