

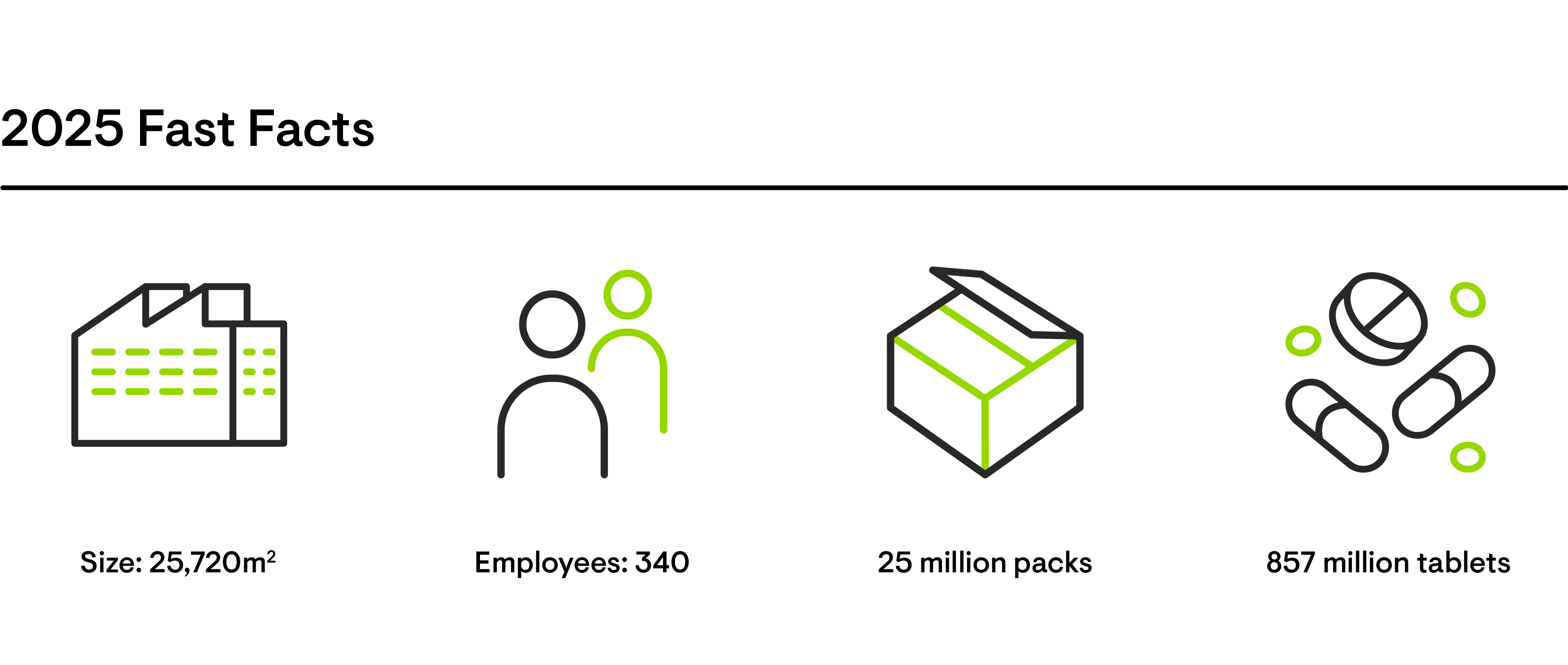
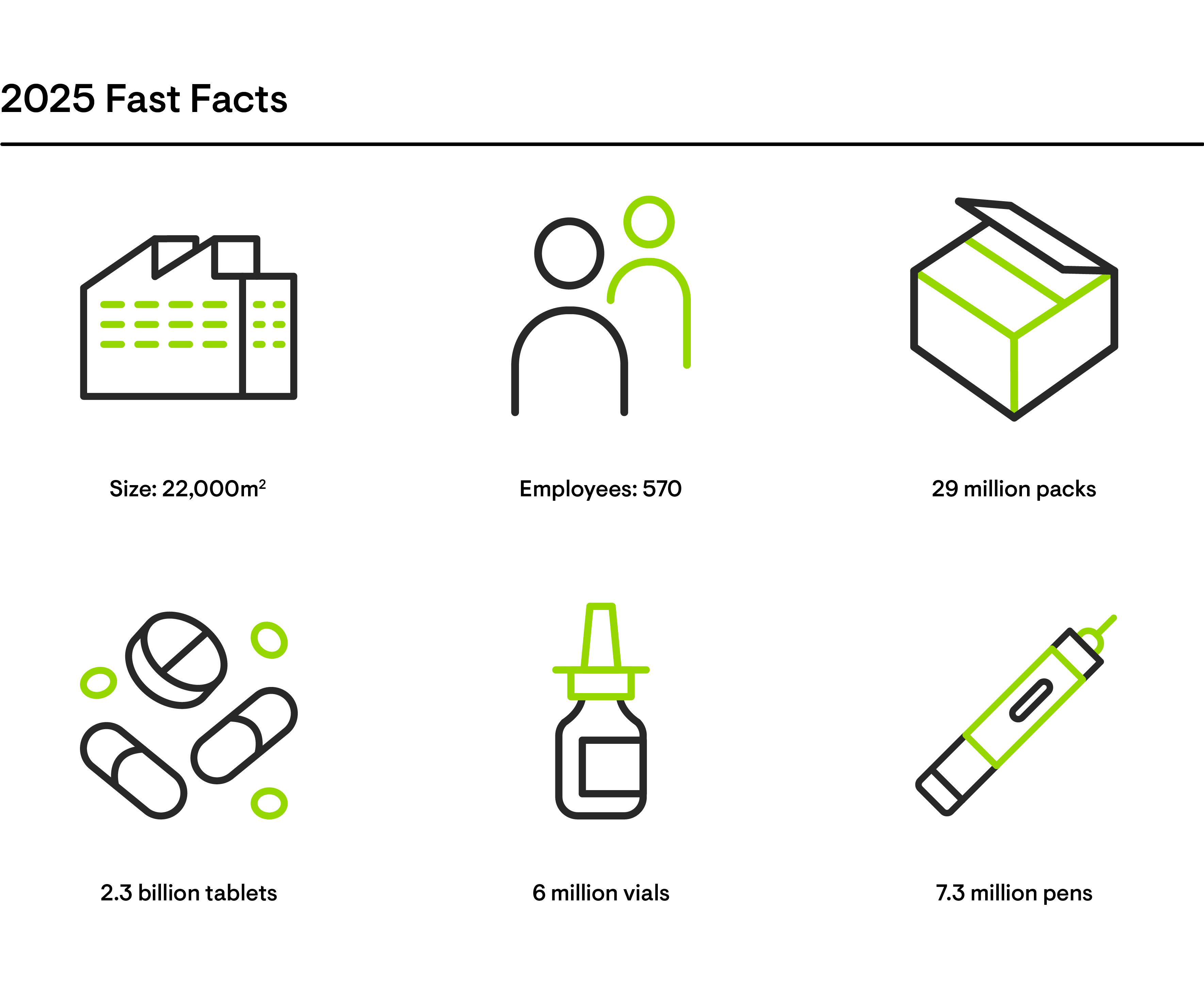
Excellence along the entire value chain
Grünenthal covers the entire value chain, from target identification, through market authorisation, manufacturing and distribution. This end-to-end approach ensures a safe, efficient and reliable product supply to patients.
Our Global Operations team aims to drive growth by ensuring excellence in their processes and by embracing digitalisation and continuously innovating how we operate. Around 2,000 people are involved in the full end-to-end value chain management of our product supply. They are committed to satisfying our customers’ demands and offering a high level of service while ensuring quality to the highest standards.
At our five specialised production sites in Chile, Ecuador, Germany, Italy and Switzerland, we manufacture our products and support our external customers. Third party manufacturing accounts for 50 percent of our overall production volume.
As a truly global partner, we know how to bring products to market at a competitive cost, with current deliveries spanning well over 100 countries worldwide. Grünenthal PRO, Grünenthal's Contract Manufacturing Business, offers a unique combination of efficient production, with high standards of quality and service level. We are able to adapt and segment our operation to different business models, from high volume generics to high complexity portfolio-driven business models, as well as continuous improvement frameworks for the competitive biopharma market.
As a well-established company, it is our long-term commitment that drives sustainable partnerships with our customers.
Our World Class Manufacturing Facilities
Santiago, Chile
Our Santiago site continues to be a centre of excellence for hormone production and the manufacturing and packaging of solid products for Chile and Latin America. In 2025, the site renewed its GMP certification with ANVISA and ISP; the team also advanced its EMA certification journey, strengthening the site’s strategic role in Grünenthal’s global network. In 2026, the team will focus on advancing the insourcing of Nexium™ and Cialis® into its manufacturing operations to deliver these products to patients worldwide.
Quito, Ecuador

In 2025, our Quito site received EU regulatory approval for the commercial production of Vimovo®. This achievement marks a major step forward, as it enables the site to supply up to 300 million Vimovo® tablets annually to patients in 17 European countries. Our facility in Quito remains Ecuador’s largest and most modern manufacturing plant for pharmaceuticals and it now plays a pivotal role in our global supply network.
Aachen, Germany

Aachen is Grünenthal’s largest packaging hub, responsible for a significant share of the company’s annual output. In 2025, the site delivered strong operational efficiency, driven by ongoing investments in automation and digitalization. Key innovations included the introduction of a QC release robot, zero-touch material flow, and digital packaging cards for the packaging centre. These advancements have further strengthened Aachen’s role in the network to support a wide range of core and integrated brands, including Nexium™, Vimovo®, and Qutenza®, and positioning the site for future growth.
API Aachen, Germany

Mitlödi, Switzerland

Our two API production sites in Europe ensured a reliable supply of key active pharmaceutical ingredients for Grünenthal and external partners. In 2025, the site focused on technical transfers, ramping up new APIs, and industrializing Rosuvastatin. Sustainability efforts advanced with the installation of heat pumps in Mitlödi, delivering CO2-free thermal energy powered by electricity from 100% hydropower.
Origgio, Italy
Origgio continues to strengthen its role in Grünenthal’s network and is entering a new growth phase. The site’s expansion master plan is well underway, with the canteen completed and construction of the new office building, vertical warehouse, and expanded packaging areas progressing on schedule. In 2025, Origgio completed the integration of the full Zomig portfolio (including Zomig ODT), shipped the first Qutenza batch packaged on site to the US, advanced the insourcing of Qutenza gel and Nexium tablets production, and started the integration of additional strategic products.





