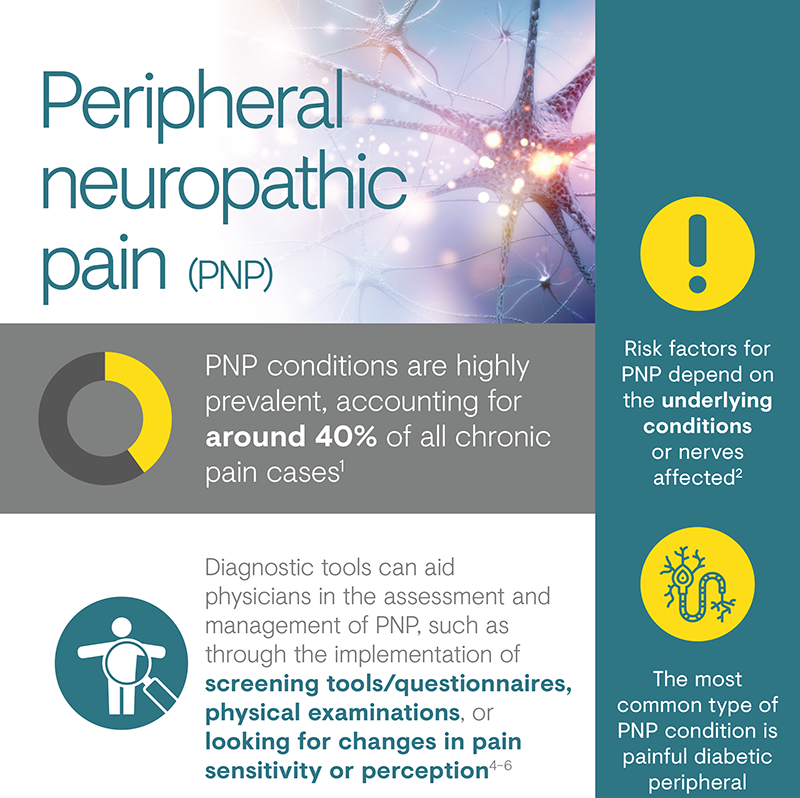
Peripheral neuropathic pain
Many chronic conditions are challenging to manage. Here we focus on key pain conditions with a high unmet need among patients and/or healthcare professionals.
Our aim here is to provide the pain community with in-depth information about these conditions; from disease understanding to management approaches.
Peripheral neuropathic pain (PNP) is a particularly distressing chronic pain syndrome that can develop after a lesion or disease affecting the peripheral somatosensory nervous system, or induced by drugs and other triggers.1,2 The causes of PNP are diverse and can be classified according to whether nerve damage is due to a systemic process, such as that caused by disease, toxins or drugs, or due to a local secondary effect of trauma, surgery or cancer.1
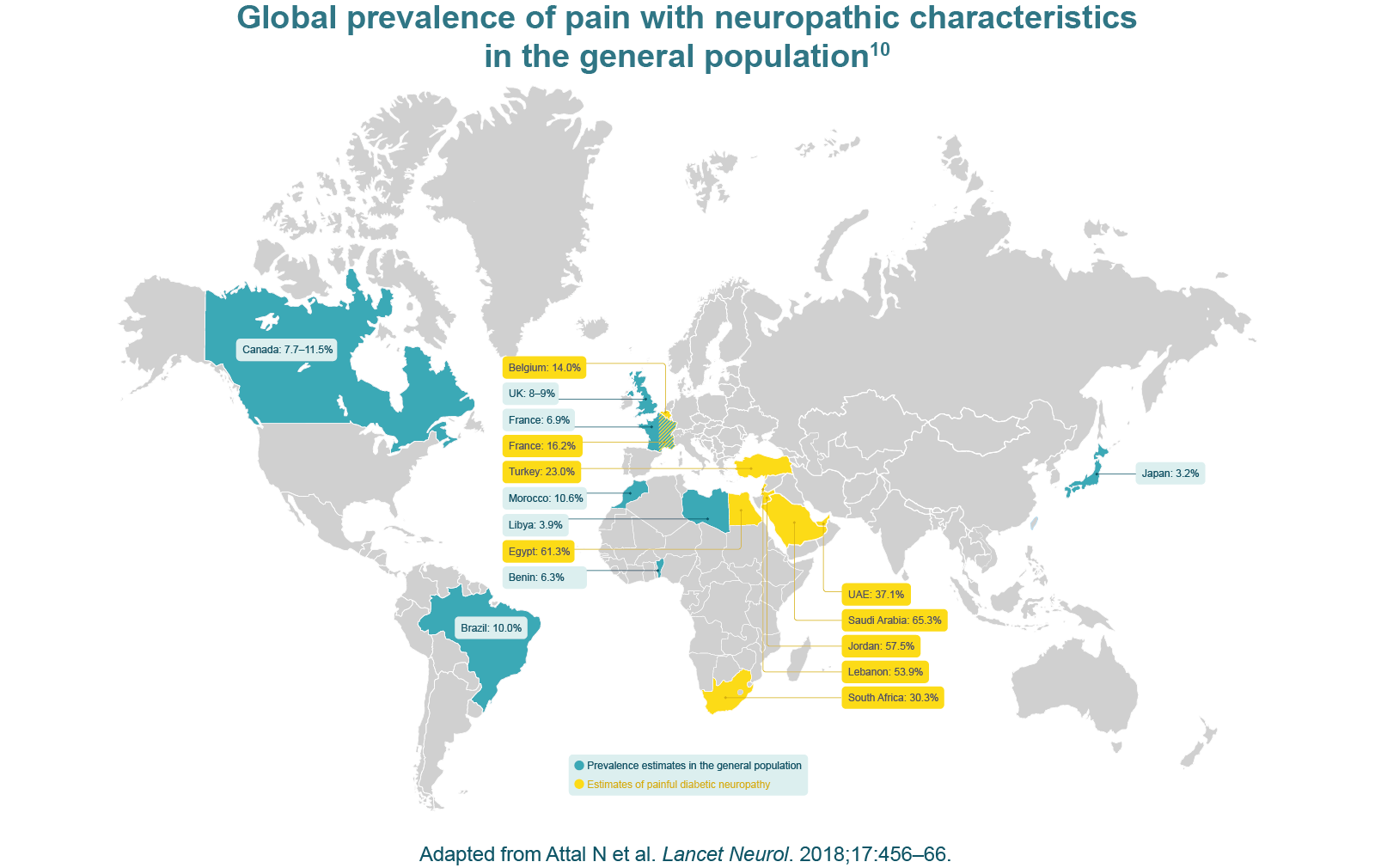
Chronic pain with neuropathic characteristics is estimated to affect 7–10% of the general population.6 Multiple causes of PNP have been described and its incidence is likely to increase owing to the ageing global population, increased incidence of diabetes mellitus and improved survival from cancer.6 A 2014 systematic review of epidemiological studies estimated the global incidence of two key PNP conditions – post-herpetic neuralgia and diabetic peripheral neuropathy – as 3.9‒42.0 per 100,000 person-years and 15.3–72.3 per 100,000 person-years, respectively.9
Chronic neuropathic pain is more frequent in women (8.0% vs 5.7% in men) and in patients over the age of 50 years (8.9% vs 5.6% in those under 50 years), and most commonly affects the lower back and lower limbs, neck and upper limbs.6
There are several types of peripheral neuropathy and the risk factors associated with each will depend on the function of the underlying condition or nerves affected. PNP can either be inherited or develop as a result of injury or illness.4 The most common type of peripheral neuropathy occurs as a complication of diabetes, affecting 60–70% of patients with diabetes.4
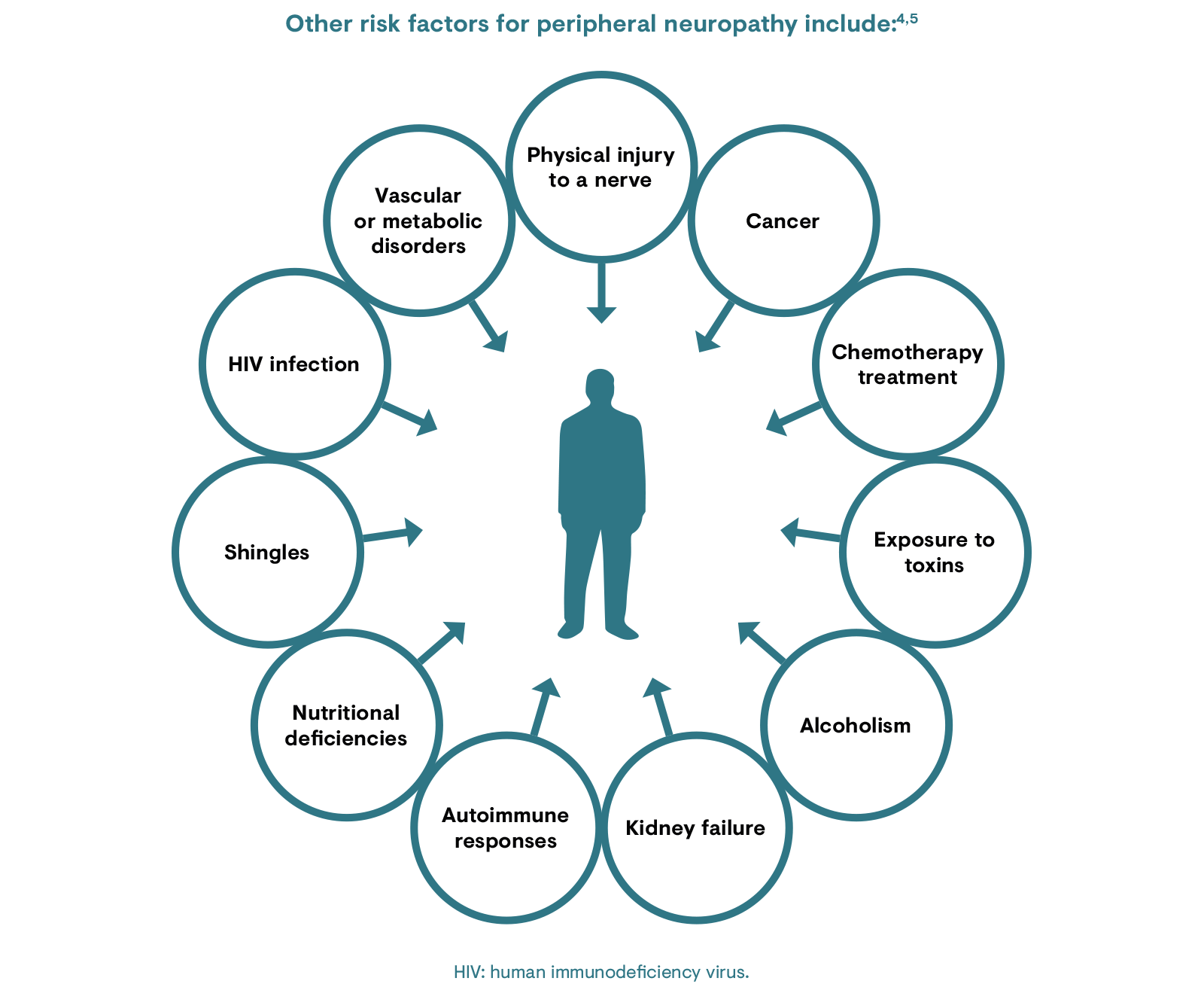
Neuropathic pain syndromes are chronic pain disorders caused as a direct consequence of a lesion or by disease of the parts of the nervous system that normally signal pain.5 They comprise a group of heterogeneous conditions, which may be caused by health conditions, certain medications or trauma. However, in certain cases, no cause can be identified and this is known as idiopathic neuropathy.11

Signs and symptoms of PNP may vary as the disease progresses and with the mood and anxiety levels of the patient.12 Neuralgias have characteristic episodic paroxysms, although other pains in neuropathy may appear as isolated attacks, or attacks of increasing intensity superimposed on a continuous pain.12
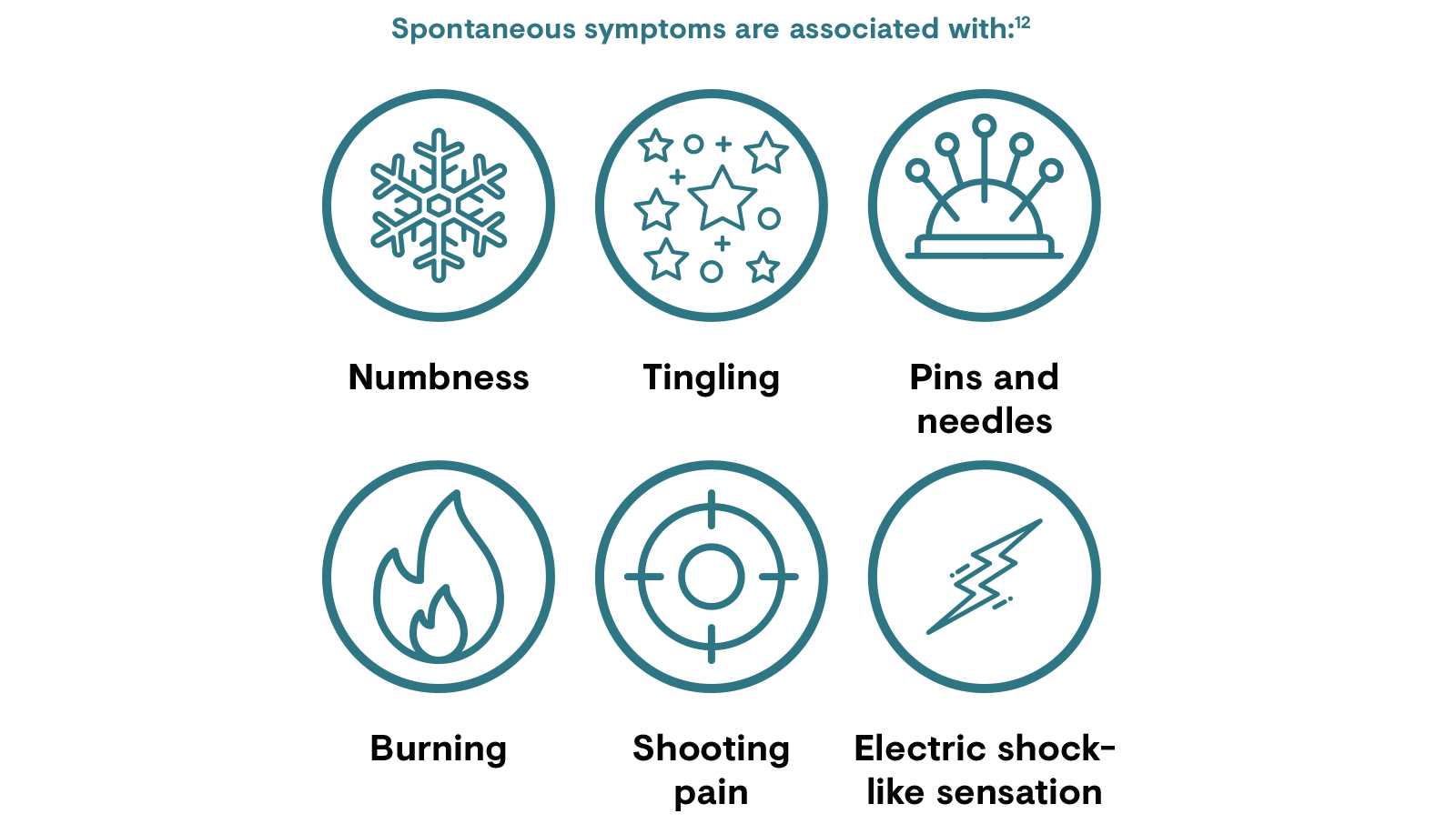
Patients may also have stimulus-dependent pain, such as allodynia (a painful response to a normally non-noxious stimuli) or hyperalgesia (increased sensitivity to pain).12
The development of neuropathic pain is multifactorial and the mechanisms are not disease-dependent. Different PNP conditions may involve the same mechanism and the same PNP symptom may be caused by different mechanisms.13
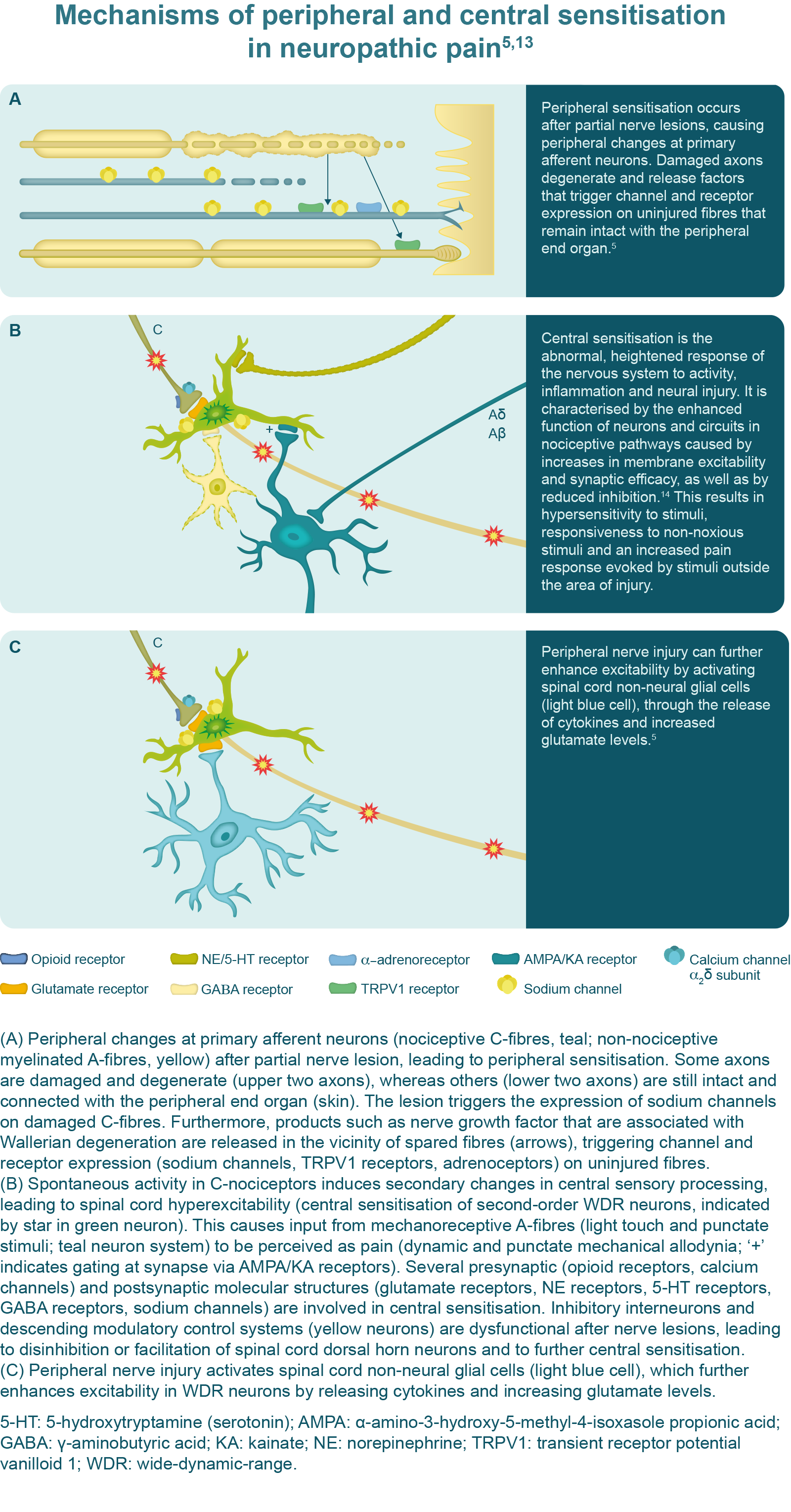
Typically, neuropathic pain is elicited by a nerve lesion of afferent pathways.13 Under healthy conditions, the activation of unmyelinated (C-fibre) and thinly myelinated (Aδ-fibre) primary afferent neurons occurs only in response to tissue damage; however, following a peripheral nerve lesion, the activity of these neurons is spontaneous and evident in both injured and adjacent non-injured neurons.5,13 It is thought that an increased expression of sodium channels in these fibres lowers the action potential threshold, causing them to fire in response to non-noxious stimuli.13 Nerve injury also leads to increased membrane excitability of nociceptive nerves and increases the expression of various receptor proteins thought to be associated with abnormal responses to thermal and mechanical stimuli.13
Allodynia and hyperalgesia in the area of the peripheral nerve lesion is caused by central sensitisation, which involves the central nervous system. Persistent firing of the peripheral nerve fibres leads to the release of excitatory chemicals within the dorsal horn of the spinal cord, which induce changes in second-order neurons, causing hyperexcitability and lowering the threshold for activation.13
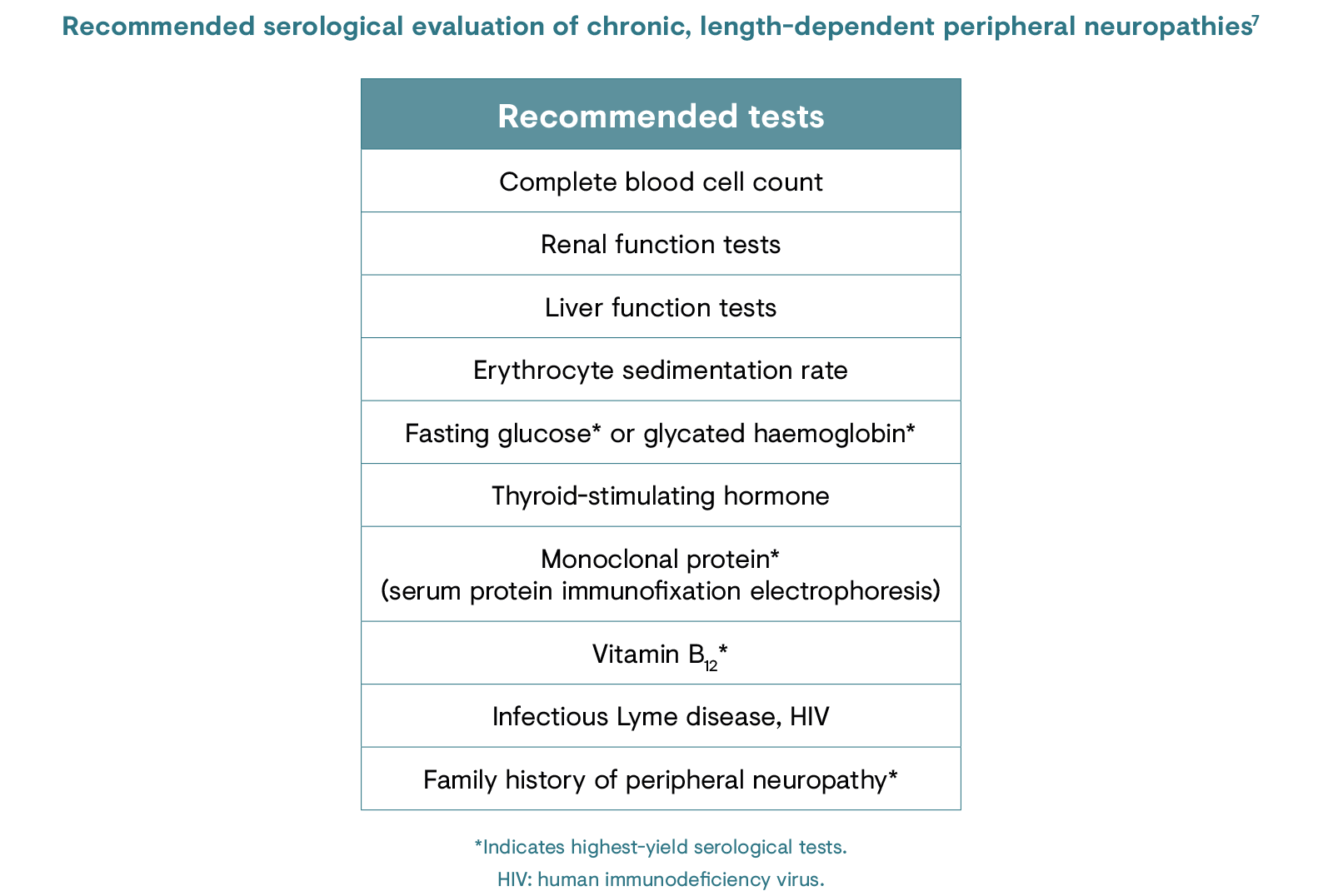
Identifying PNP conditions in patient subgroups in which the disorder is highly prevalent (e.g. diabetic peripheral neuropathy in individuals with diabetes) involves regular screening and physical examination.7
In-office screening tools include questionnaires designed to test for symptoms of PNP, such as the Neuropathic Pain Symptom Inventory (NPSI), the Neuropathic Pain 4 Questions (DN4; Douleur Neuropathique en 4 Questions), the Leeds Assessment of Neuropathic Symptoms and Signs (LANSS), the localised neuropathic pain (LNP) screening tool, as well as the painDETECT screening questionnaire, which can be used to identify neuropathic components in patients with back pain.6,8,15
Physical examination for PNP in the primary care setting typically involves observing light touch perception to a monofilament (to demonstrate allodynia), superficial pain perception to a pinprick (to demonstrate hyperalgesia) or testing of ankle deep-tendon reflexes (absent reflexes may indicate peripheral neuropathy).6,7,16
For practical guidance on how to conduct a physical examination, watch the following videos:
If a patient presents with pain of an unknown cause, as well as considering the patient’s history and anatomy, screening questionnaires and physical examination, as described above, can facilitate diagnosis of PNP.
The LNP screening tool was developed by the Neuropathic Pain Special Interest Group of the International Association for the Study of Pain to facilitate easy and reliable diagnosis of LNP.8 It consists of four steps: assessment of the patient’s pain history, assessment of pain distribution, sensory examination (inside and outside of the painful area) and assessment of the size of the painful area.
Various healthcare professionals are involved in the diagnosis and treatment of PNP conditions. General practitioners (GPs) are considered the ‘gatekeepers’ of the healthcare system and diagnose most cases of PNP.8 If a GP makes a diagnosis of PNP, treatment can be initiated. If there is diagnostic uncertainty or if treatment is not sufficient, then patients are generally referred to a pain clinic where they may see a pain specialist, neurologist or neurosurgeon, rehabilitation physician and/or physiotherapist for more advanced pharmacological or surgical management of neuropathic pain.
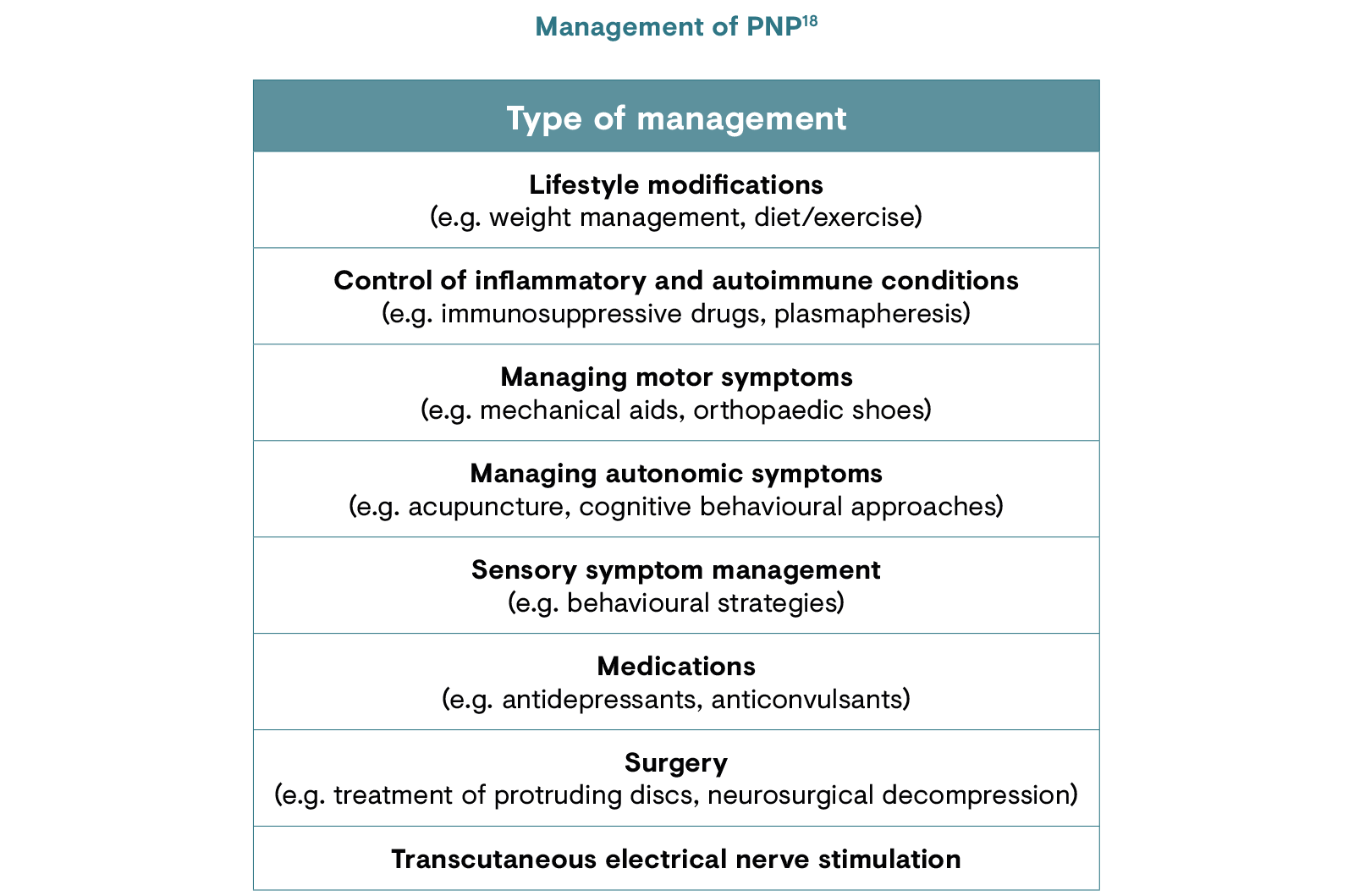
Treatment for PNP varies depending on the type of nerve damage that has occurred, the symptoms and the location of pain. Currently used pharmacotherapies are aimed at the mechanisms of neuropathic pain, but these mechanisms are complex, often overlapping and incompletely understood.13,17 The general approach is a stepwise process to identify which drugs or drug combinations provide the greatest pain relief with the fewest side effects.13 The main goal of pharmacotherapeutic management of PNP is a clinically important reduction in pain: moderate relief is defined as a 30% reduction in pain while substantial relief is defined as a 50% reduction in pain.17
With proper understanding, some patients may be able to reduce or even eliminate their need for medications, and functional recovery may be possible with adequate management as long as the nerve cell itself has not died.18
Click the links to view the current treatment guidelines from the European Federation of Neurological Societies (EFNS) and the Neuropathic Pain Special Interest Group (NeuPSIG).
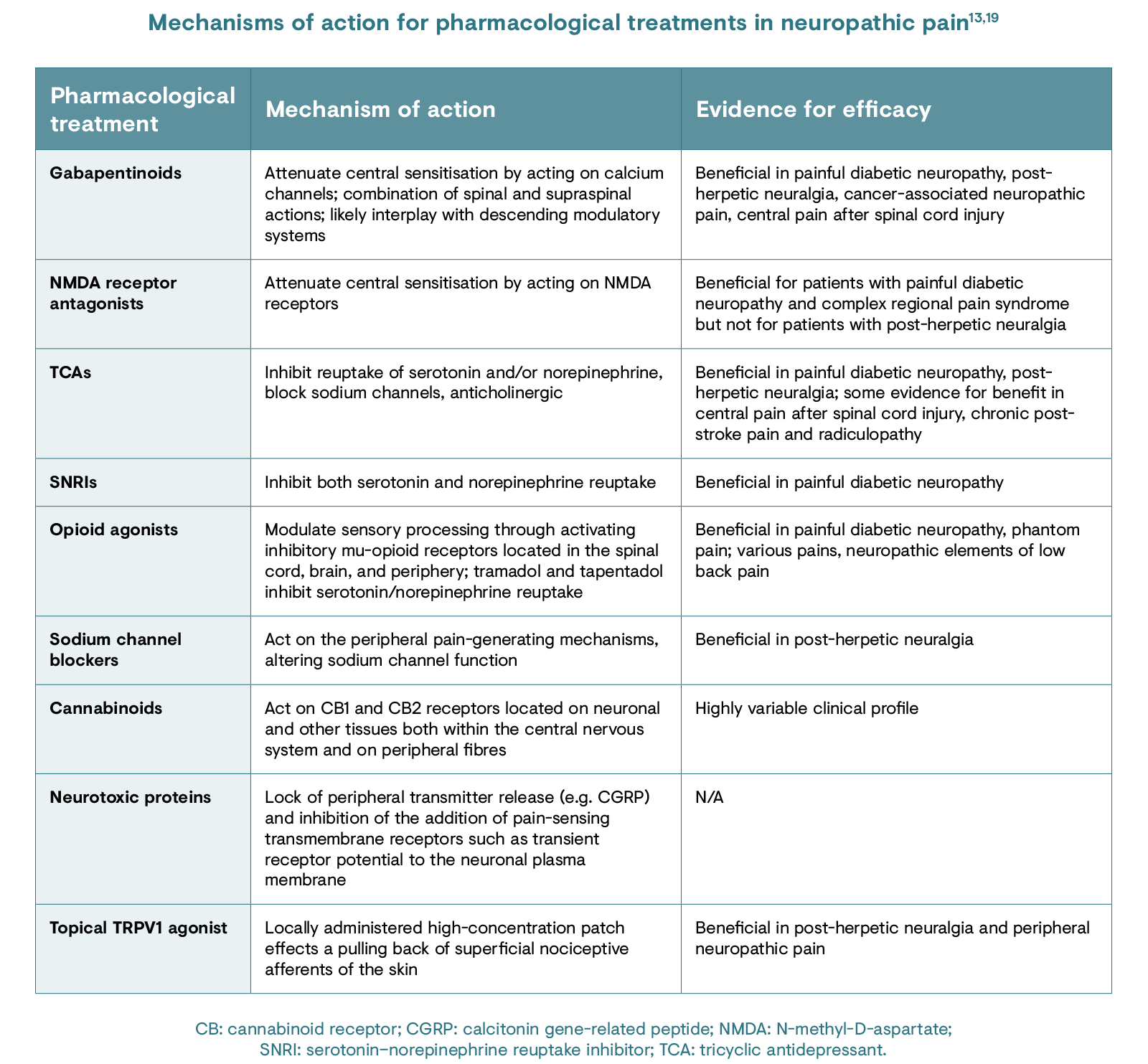
Understanding the mechanisms responsible for the pathogenesis of neuropathic pain in the individual can allow for a targeted treatment approach.19 The table below describes the different available treatments for neuropathic pain and their mechanisms of action.13,19 However, there is uncertainty within the scientific community regarding the central pain-generating mechanisms of PNP and its implications for treatments. 20
Despite advances in understanding the complex neurobiology of pain, the clinical management of neuropathic pain is often inadequate, with several studies citing ineffective management approaches and prescription patterns.21 There is also a paucity of novel drugs and treatment approaches; several promising drugs have failed in late-stage development.2
Inadequate diagnoses may play a role in the ineffective treatment of neuropathic pain. For instance, a UK study found that 79% of patients had suffered pain for more than a year before referral to a pain clinic.21 Behavioural and psychiatric comorbidities, such as depression, anxiety, sleep disorders and substance abuse, may be a consequence of delayed diagnosis or mistreatment.21
1. Colvin LA & Dougherty PM. Br J Anaesth. 2015;114:361–3.
2. Baron R et al. Pain. 2017;158:261–72.
3. Sicras-Mainar A et al. J Comp Eff Res. 2018;7(7):615–25.
4. The Foundation for Peripheral Neuropathy. What is peripheral neuropathy? 2016. Available at: https://www.foundationforpn.org/what-is-peripheral-neuropathy/types-risk-factors. Accessed May 2020.
5. Baron R. Nat Clin Pract Neurol. 2006;2:95–106.
6. Colloca L et al. Nat Rev Dis Primers. 2017;3:17002.
7. Watson JC & Dyck PJ. Mayo Clin Proc. 2015;90:940–51.
8. Mick G et al. Curr Med Res Opin. 2014;30:1357–66.
9. van Hecke O et al. Pain. 2014;155:654–62.
10. Attal N et al. Lancet Neurol. 2018;17:456–66.
11. National Health Service. Peripheral neuropathy causes. 2019. Available at: https://www.nhs.uk/conditions/peripheral-neuropathy/causes/. Accessed May 2020.
12. Marchettini P et al. Curr Neuropharmacol. 2006;4(3):175–81.
13. Baron R et al. Lancet Neurol. 2010;9:807‒19.
14. Latremoliere A & Woolf CJ. J Pain. 2009;10(9):895–926.
15. Freynhagen R et al. Curr Med Res Opin. 2006;22(10):1911–20.
16. Walker HK. Chapter 72: Deep tendon reflexes. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition. Boston: Butterworths; 1990. Available at: https://www.ncbi.nlm.nih.gov/books/NBK396/. Accessed May 2020.
17. Brooks KG & Kessler TL. Treatments for neuropathic pain. In: Clinical Pharmacist. 2017. Available at: https://www.pharmaceutical-journal.com/research/review-article/treatments-for-neuropathic-pain/20203641.article. Accessed May 2020.
18. National Institute of Neurological Disorders and Stroke. Peripheral neuropathy fact sheet. 2020. Available at: https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Peripheral-Neuropathy-Fact-Sheet. Accessed May 2020.
19. Bannister K et al. Annu Rev Pharmacol Toxicol. 2020;60:257–74.
20. Meacham K et al. Curr Pain Headache Rep. 2017;21:28.
21. Harden N & Cohen M. J Pain Symptom Manage. 2003;25(Suppl 5):S12–17.